It’s Friday the 13th — a day some consider ominous, but not Lupin Managing Director Nilesh Gupta. Dressed in a traditional black kurta, he walks in for our meeting on the ninth floor of the company’s Mumbai headquarters — where he and his sister Vinita, the Chief Executive Officer, have their offices — to discuss the road ahead for the estimated ₹22,707-crore home-grown multinational drugmaker.
Nilesh is reminded that September will mark 13 years since he and his sister assumed their leadership roles. Good-humouredly he responds that the number (13) has been lucky for him — his surname, too, put him thereabouts in the alphabetical roll-call in class.
New Lupin Research Park facility, which was inaugurated by its late founder, Desh Bandhu Gupta, in 2017
The silence across the floor, and inside Lupin’s meeting room, lined outside by plants, belies a reality that is giving several corporate chiefs sleepless nights. A war rages in West Asia, supply chains are being disrupted again (after the Covid pandemic), and shortages begin to cast a long shadow — even in a country like India, which has a thriving domestic pharmaceutical industry.
Lupin has navigated tough times in the past too, under the stewardship of its founder, the late Desh Bandhu Gupta (he passed away in June 2017). DBG’s (as he was called) children now steer the ship. Is their strategy on handling global challenges, this time posed by a war, different from their father’s?
DBG would have taken bigger bets and bolder decisions, says Nilesh, because he was faced with “existential challenges”. “We’ve not dealt with existential concerns, Vinita and me… I do think we stand on the shoulders of giants,” he says, adding that the industry will overcome the current crisis.

Lupin’s Pithampur plant houses the world’s highest capacity metered-dose inhaler (MDI) line
| Photo Credit: Devendra Purbiya
Framing the big picture for Lupin, Nilesh says innovation is the space for their big bets for the future. The earlier vision for a new therapy every year, set by DBG, is done, says Nilesh. “We’re into everything as far as generics… therapy-wise, dosage form-wise… that was the right strategy at that point of time,” he says.
The journey continued with the making of dermatology products, oral contraceptives, ophthalmology products and inhalation formulations. And through it, Lupin evolved as a producer of complex generics — products that few could make and, hence, marked by less competition and better prospects. Then came biosimilars: “we see biosimilars being 50 per cent of our European market, for example… and a good part of the rest would be inhalation,” says Nilesh. (Biosimilars, which are large complex molecules, are defined as “highly similar” to biological products.)

Lupin’s plant in Nagpur
Lupin played a critical role in making cephalosporins and tuberculosis drugs, for example. “Now it’s going to be about innovation,” says Nilesh.
Explaining Lupin’s entry into diagnostics, digital support and a neurological rehabilitation initiative, he says “the combination of diagnosis and pharmaceuticals seemed like a good place to start”, and the digital health division opened up a different avenue for cardiac treatment and support.
As the three grow, Lupin becomes a “broader healthcare player”, he says.
Crucial US market
Lupin’s growth plans include acquisition, says Nilesh, adding, “Our acquisition priorities are speciality in the US and Rx (prescription products) in India… OTC (over-the-counter products) is definitely a third… Vitabiotics is interesting because it’s, again, OTX (prescribed, but non-prescription product).” Lupin was reported to be evaluating UK’s Vitabiotics, a nutraceutical company.
Outlining the importance of the US market, Vinita says, “The US is, and will continue to be a cornerstone of Lupin’s global strategy.” It’s an important contributor to Lupin’s financial performance, she says, pointing out that Lupin is the third-largest generic pharmaceutical company in the US by prescription volume — contributing to the health of Americans, besides generating considerable savings to the US healthcare system.
DBG’s recently launched biography captures a time when he asked a young Vinita to set up and establish Lupin in the US. In the 1990s, the generics market looked very different compared with today, says Vinita. The Hatch-Waxman Act of 1984, in simplifying the Food and Drug Administration (FDA) approval process for generics and granting a 180-day exclusivity period for first-to-file companies, created the right conditions for growth, she adds. Being a part of the industry’s transformation and driving the company’s expansion in a strategically important market was exciting and energising, she says, adding, “It ultimately laid the foundation for the global pharmaceutical company we are today.”
The US generics landscape changed and Lupin sharpened its strategic focus on complex generics, biosimilars and speciality products, which can sustain long-term business and mitigate the impact of external pricing pressures, she says.
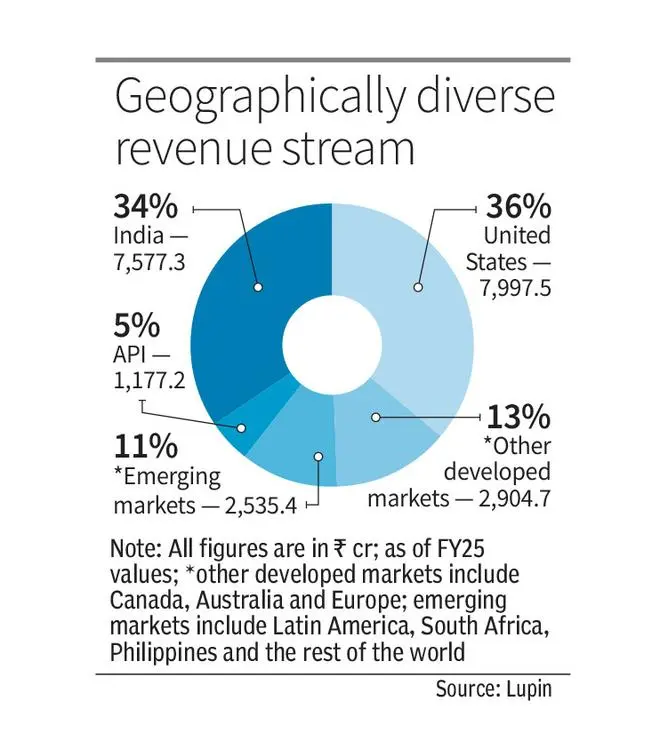
Alongside the US, home market India is central to the company’s strategy, as also Europe — another growth driver, with complex generics like respiratory products and biosimilars, she says.
Bold bets
Being a large generic player does not necessarily equip a company to be a large innovation player, as the capabilities are different, and not necessarily India’s strong point, Nilesh says. However, India can be leveraged for those capabilities, he adds. “We’re now starting to operate innovation at two levels — one for the world, and the other for India.”
In India, it could be cardiac, diabetes, respiratory and oncology molecules — all as new chemical entities (NCEs). “But the cost of development is lower for just India as a market. The time-to-market is shorter. And, therefore, you can have much more shots on goal. Obviously, you’ll still have failure because you’re taking NCE risk eventually,” he explains.
The expectation is to have “a bunch of products in clinical trials” — for the first time, NCEs for India, he says, on picking up early innovation from China and Korea for development. “We are biting much deeper on the innovation side… on technology,” he says, pointing to AI and innovations to manufacture for the future, and building into it compliance.
Going back to the backdrop of the war, Nilesh points to the opportunity to “scale even more elsewhere in the world”. But aren’t there price increases, shortages? “Hundred per cent, because people use this to their advantage,” he says, citing increasing logistics and insurance costs. These are short-term, they will settle down, he adds. “I would by no means make light of the misfortunes of war,” he says, but looking ahead, it’s got to be innovation at scale — “much bigger bites”.
More Like This
Published on March 30, 2026